The sudden death of a stallion is a devastating event, not just emotionally, but potentially financially as well. In addition to any competitive goals the stallion was working towards, his untimely death may mean the loss of future breedings, particularly if a stock of frozen semen was not banked up whilst he was alive. In this situation it may be possible to harvest and freeze epididymal sperm by castrating the stallion immediately after death or euthanasia. Alternatively, collection and freezing of epididymal sperm may be an elective procedure performed at the time of castration. This offers an advantage to young stallions, i.e. some semen can be frozen without taking time out from a busy training schedule, or without exposing a young stallion to the breeding environment. For the mature stallion it represents an additional opportunity to collect semen for freezing. Here we review the process of epididymal sperm harvesting for freezing, and its subsequent application for breeding purposes.
What is the Epididymis?
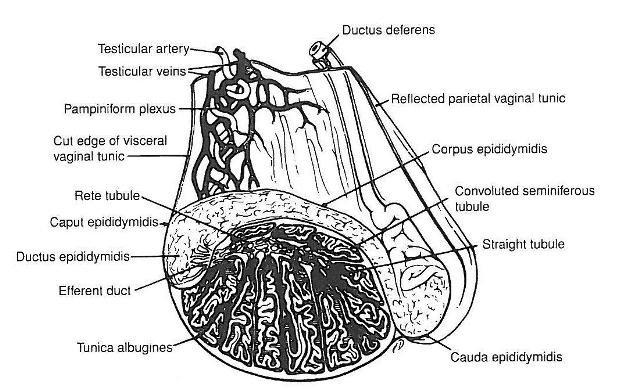 The epididymis can be thought of as a storage organ for sperm before ejaculation and it is comprised of three sections – the caput (head), corpus (body) and cauda (tail). As you can see in the figure 1 (right) the epididymis lies atop the testicle; the caput is relatively flat, it has a “J” shape and is closely attached the front of the testicle, the corpus is a cylindrical structure loosely attached to the dorsal surface of the testicle, and the cauda epididymis is large and bulbous, loosely attached to the back of the testicle. The efferent ducts of the testis feed into the proximal caput epididymis and converge to form a single duct, the epididymal duct, which continues throughout the caput, corpus and cauda epididymis. The epididymal duct may be 70-80m long and follows a tortuous pattern of pleats and folds, particularly within the cauda epididymis, where it then exits and becomes continuous with the deferent duct, or vas deferens, that proceeds up through the spermatic cord to the pelvic urethra.
The epididymis can be thought of as a storage organ for sperm before ejaculation and it is comprised of three sections – the caput (head), corpus (body) and cauda (tail). As you can see in the figure 1 (right) the epididymis lies atop the testicle; the caput is relatively flat, it has a “J” shape and is closely attached the front of the testicle, the corpus is a cylindrical structure loosely attached to the dorsal surface of the testicle, and the cauda epididymis is large and bulbous, loosely attached to the back of the testicle. The efferent ducts of the testis feed into the proximal caput epididymis and converge to form a single duct, the epididymal duct, which continues throughout the caput, corpus and cauda epididymis. The epididymal duct may be 70-80m long and follows a tortuous pattern of pleats and folds, particularly within the cauda epididymis, where it then exits and becomes continuous with the deferent duct, or vas deferens, that proceeds up through the spermatic cord to the pelvic urethra.
Transit time for spermatozoa through the epididymis is about 8-11 days, during this time they undergo significant maturation. As they pass through the epididymis fluid is resorbed and the epididymis also provides its own secretions, influencing the membrane properties of the spermatozoa. Sperm that leave the testis are immotile and incapable of fertilization, however by the time they reach the tail of the epididymis they have achieved the ability to fertilize. Approximately 60% of spermatozoa in the excurrent duct system (i.e. from the exit of the testis on through the vas deferens) are in the cauda epididymis; therefore this is a significant reservoir of potentially fertile sperm that could be harvested.
Preparing the Testicles for Epididymal Harvest
Generally the timing of euthanasia is often unexpected and the location may not be suitable for subsequent post-mortem tissue processing or semen freezing. If the stallion cannot be transported to a suitable processing center prior to castration or euthanasia, then it will be necessary to transport the testicles to a processing facility. Although this is a stressful time it is important to be prepared and to communicate with the processing center, ideally before euthanasia and/or castration, so specific instructions can be obtained and they can be prepared to receive and process the tissues. Appropriate removal of the testicles, packaging and transport can have a big influence on the outcome of the procedure, so it is important to optimize this process as much as possible by being prepared.
In general, both testes and associated epididymides are removed using standard surgical castration procedures. Care should be taken to leave the tail of the epididymis and as much of the vas deferens associated with each testis intact. Ideally the vas deferens should be transected as high as possible and tied off to prevent the loss of sperm. Each testis and epididymides should be rinsed with saline and placed in a plastic bag or rectal sleeve. For transport the testes should be kept chilled, packed in an insulated shipping container with ice packs (do not use dry ice). An ideal transport vessel is the Equitainer used for shipping cooled semen. Just as you would for cooled semen shipments, place the coolant cans in the bottom of the Equitainer and then the bag containing the testicles on top. Whether using an Equitainer or alternative shipping box, wrap the bag of testicles in a towel so they are not in immediate contact with the ice packs, thereby preventing freezing of the tissue. Most processing centers would prefer to receive the testicles as soon as possible, ideally within 24hrs of castration. If same-day shipment through counter to counter airline service is an available option it could be utilized to reduce transit time.
Another advantage of communicating with the processing center prior to euthanasia or castration is to review the stallion’s health testing status. Some storage and distribution centers require proof of EVA status prior to storage and distributing frozen semen. Standard bio-security protocol should be to pull blood from the donor stallion for Equine Infectious Anemia (EIA) and Equine Viral Arteritis (EVA) testing as a minimum, and ideally to take swabs of the urethra and urethral fossa for Contagious Equine Metritis (CEM) testing. These samples can be procured prior to euthanasia or at the time of castration. This may prevent unexpected restrictions on semen storage and distribution after processing, and potential disease outbreak if contaminated semen is used in a breeding program. It is responsible practice for a stallion owner to only utilize semen of known health status in a commercial breeding program.
Harvesting of Sperm from the Epididymis
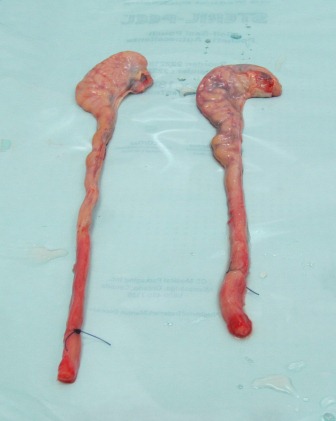 Upon arrival at the processing center, the epididymis should be dissected free from its attachment to the testicle. The cauda, or tail of the epididymis and the connected vas deferens are then dissected from the rest of the epididymis (i.e. the corpus and caput), see photo left.
Upon arrival at the processing center, the epididymis should be dissected free from its attachment to the testicle. The cauda, or tail of the epididymis and the connected vas deferens are then dissected from the rest of the epididymis (i.e. the corpus and caput), see photo left.
There are two basic methods for extracting sperm from the epididymis, either by retrograde flushing of the sperm through the epididymis or by utilization of the “float-up” method. In the latter method, the vas deferens and cauda epididymis are placed in petri dish (or embryo search dish), with semen extender or appropriate dilution media. The tissue is incised in multiple locations and the dish agitated to encourage release of the sperm from the tissue into the surrounding media. At SBS we prefer the flush method, primarily because more sperm are generally obtained through retrograde flushing than by the float up method. It also provides a more concentrated volume of sperm cells, therefore less potential for contamination with blood or tissue. For the flush method the lumen of the vas deferens is canulated and a syringe containing semen extender (or an appropriate dilution media) is inserted. The epididymis is cut close to the corpus-cauda junction to allow sperm to exit. The tissue is then suspended over a collection tube and the syringe slowly and carefully depressed to flush the sperm through the cauda epididymis.
Cryopreservation of Epididymal Sperm
After the sperm have been harvested from both epididimides the semen solutions are combined and the sperm concentration is determined in order to calculate yield. Depending upon the concentration or the method employed for harvesting centrifugation may be required. For example, a basic skim milk glucose extender may be used for the float or flush method. If this is the case, the sperm must then be centrifuged and resuspended in a suitable cryopreservation extender. Alternatively, if a large volume of flush media was used or sperm recovery was unexpectedly low, resulting in a low sperm concentration, then centrifugation may be required in order to concentrate the cells appropriately for semen freezing. An advantage of the flush method is that the cryopreservation extender can be used as the flush media, thereby eliminating the requirement for centrifugation.
Once resuspended in the freezing extender the cryopreservation process continues just the same as for ejaculated spermatozoa, i.e. the semen is loaded into 0.5mL straws and frozen by utilization of a programmable cell freezer machine or passive vapor freezing system. A disadvantage of freezing sperm after epididymal harvest is that semen from the stallion may not have been frozen before. Consequently how the stallion’s sperm will respond to the semen freezing process is unknown and the standard “test freeze” on the first ejaculate, used to identify the freezing extender that works best for that particular stallion, may not have been performed. Nonetheless a suitable freezing extender can be selected that offers the best chance of success, it may not be the optimal choice for that particular stallion but usually satisfactory results can be obtained.
How many Doses can I Expect to Obtain?
The number of sperm that can be recovered from the paired epididymides of a stallion is typically more than a regular ejaculate from the same stallion. The two caudal epididymides of a normal, sexually rested stallion should contain about 50 billion sperm. However, sperm production is highly variable between stallions and is directly related to testicular size. Consequently, a stallion that normally produces 4-5 billion sperm in an ejaculated sample is likely to have less sperm stored in an epididymis than a stallion that typically ejaculates 10-12 billion sperm. Ejaculation frequency prior to harvesting can also influence total sperm numbers recovered from the epididymis, e.g. a stallion collected on a daily basis prior to epididymal harvest would have less sperm in the epididymial reservoir than if he had been sexually rested. Therefore, you could expect a range of 5 to 50 breeding doses obtained from a pair of testes.
The quality and motility of sperm obtained after epididymal harvest is usually good for reproductively normal stallions, but is naturally dependent upon the inherent sperm quality for that particular stallion. For example, the procedure is less likely to be successful if the stallion has had a prolonged illness or suffered an age-related reduction in semen quality and/or fertility before death. The success of the procedure can also be influenced by the temperature and time of transit to the processing center, in addition to the skill of the technician preparing the tissue and harvesting the sperm. The success of cryopreservation is in turn influenced by the skill of the technician, the equipment and technology used for processing and upon how well the stallion’s sperm responds to semen processing and freezing.
Fertility of Epididymal Sperm
The first reported pregnancy with frozen-thawed stallion sperm was actually achieved with epididymal sperm in 1957. Pregnancies have since been confirmed in frozen thawed epididymal sperm harvested, processed and inseminated on site, or following cooled transport of the testis to a separate location for processing and subsequent shipping of the frozen semen to the mare for insemination. However, the fertility of frozen-thawed epididymal sperm is generally believed to be less than frozen and thawed ejaculated sperm. The main reason for this is that spermatozoa are undergoing constant maturation as they pass through the epipidymis, this maturation continues and is influenced by exposure to the seminal plasma at the time of ejaculation; furthermore maturation continues after introduction and interaction with the female reproductive tract. Extraction of epididymal sperm essentially eliminates the opportunity for spermatozoa to be influenced by the seminal plasma at the time of ejaculation. To compensate for this seminal plasma is often added back to epididymal sperm prior to cryopreservation, or at the time of insemination. The pregnancy rate with frozen-thawed epididymal sperm may be optimized by use of assisted reproductive techniques such as low dose rectally guided or hysteroscopic deep uterine horn insemination or intracytomplasmic sperm injection (ICSI).
Addition of Seminal Plasma
The proposed benefits of adding seminal plasma to epididymal sperm are as follows:
- Sperm harvested from the epididymis miss the opportunity of being exposed to seminal plasma at the time of ejaculation and their maturation may therefore be comprised.
- It is known that a small percentage of seminal plasma in the volume of semen inseminated into the mare can help mitigate the inflammatory response of the uterus to insemination.
Seminal plasma could be prepared and frozen from the same stallion, if the opportunity is available, or more commonly aliquots of seminal plasma are prepared and frozen from the ejaculate of a different stallion with good semen quality and known fertility. Seminal plasma can be added to the freezing extender prior to cryopreservation of epididymal sperm. Alternatively, seminal plasma can be used as the flush media when harvesting sperm with the retrograde flush method, followed by subsequent dilution with freezing extender. If the epididymal sperm were not frozen in the presence of seminal plasma, seminal plasma could be added to the semen after thawing, allowing a period of exposure prior to insemination. The results in the literature are inconclusive; some studies report an improvement in fertility with the addition of seminal plasma, whereas others do not. Nonetheless, it is not considered to be detrimental and therefore given the potential benefits most practitioners will add seminal plasma if available. Generally, the percentage of seminal plasma added back in these cases is approximately 5%.
If you are interested in learning more about freezing epididymal sperm or require the use of this service at any time in the future, please contact Select Breeders Service for more information (410-885-3202, info@selectbreeders.com).
Figure 1: Pickett BW et al, (1989) Management of the Stallion for Maximum Reproductive Efficiency, II. Animal Reproduction Laboratory Bulletin No. 5. Fort Collins, Colorado State University.





