The most commonly used measure of semen quality is sperm motility, specifically “progressive motility”. But what exactly does that mean and how is it determined? The minimum post-thaw “progressive motility” generally recommended for commercial distribution of frozen semen is 30% to 35%. This article will address some of the factors that can affect the assessment of sperm motility as well as the various problems associated with techniques used for determining “progressive motility”.
He Said She Said: The problem with subjective motility
One of the main problems with using motility as the basis for determining the expected or even guaranteed quality of frozen semen is the variability between technicians or laboratories in subjective estimates of sperm motility. Determining progressive motility subjectively involves visually scanning several fields of motile sperm under a microscope and estimating the percentage of the population that are moving in a progressive fashion. Experienced technicians all trained in a single laboratory can become very consistent between one another in their estimations of progressive motility, however when two technicians from different laboratories assess the same sample there can be tremendous variability in the reported values.
When a mare owner purchases frozen semen they are expecting that the quality will meet minimum standards after thawing. They expect that analysis of the semen at the time of breeding will confirm the quality that was reported by the seller or the laboratory that froze the semen. However, if those assessments are made subjectively either by the freezing lab or the veterinarian performing the AI then they may be worlds apart in their estimation of progressive motility. This can lead to problems between stallion owners and mare owners or frozen semen brokers and the purchasers of those doses of semen.
Technology to the Rescue
Several years ago when I was working with engineers and programmers at Hamilton-Thorn Research on development of computer assisted sperm motility analyzer technology (CASA) I was asked what motion parameters were important to measure for the analysis of equine sperm motility. This new technology allowed us to track sperm motion very precisely and make calculations on any number of motion parameters. I told the programmers that the percentage of “progressively motile” sperm in a sample was the standard measure of semen quality and that is what was used to calculate insemination doses. They asked me to define “progressive motility”. I replied what I had always been taught and what I had always taught others which was “the percentage of sperm moving in a relatively straight path with reasonable velocity”. “Well, how straight is relatively straight and how is that measured and what velocity is reasonable and how is that measured" was their response. So the advent of CASA required a more precise definition of what motion characteristics distinguished a progressively motile sperm from a non-progressively motile sperm. Using these objective systems we can measure the actual velocity in microns per second of hundreds of sperm in a matter of minutes.
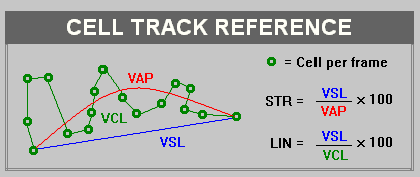 Most systems will measure two or three types of velocity. Curvilinear velocity (VCL) is the distance over time of the actual path of the sperm head whereas path velocity (VAP) is the distance along a 5 point mathematically smoothed path that the sperm travels over time. A third velocity measurement “straight line velocity” (VSL) is the straight line distance between the position of the sperm head in the first frame of analysis and the position of that sperm head in the last frame of analysis over the fixed time of analysis. We can also measure the ratio of how straight the paths of those cells are over time. These are expressed as “percent straightness” (STR) or percent linearity (LIN). STR is the ratio of the straight line velocity (VSL) to the average path velocity (VAP) expressed as a percentage and LIN is the ratio of VSL over VCL. The diagram above illustrates a theoretical sperm path and how these parameters are calculated.
Most systems will measure two or three types of velocity. Curvilinear velocity (VCL) is the distance over time of the actual path of the sperm head whereas path velocity (VAP) is the distance along a 5 point mathematically smoothed path that the sperm travels over time. A third velocity measurement “straight line velocity” (VSL) is the straight line distance between the position of the sperm head in the first frame of analysis and the position of that sperm head in the last frame of analysis over the fixed time of analysis. We can also measure the ratio of how straight the paths of those cells are over time. These are expressed as “percent straightness” (STR) or percent linearity (LIN). STR is the ratio of the straight line velocity (VSL) to the average path velocity (VAP) expressed as a percentage and LIN is the ratio of VSL over VCL. The diagram above illustrates a theoretical sperm path and how these parameters are calculated.
So when using an objective measure of sperm motility instead of a subjective one there should be no discrepancies between two labs that are both using the same CASA system in the analysis of progressive motility. Unfortunately this is not necessarily the case. The actual sperm motility can be influenced by a number of factors such as thawing rate, type of extender used to dilute the sample, incubation time and temperature, type of slide or chamber used to view the sample and temperature of the microscope stage. There are also a number of instrument related “user-defined” factors that do not affect the actual motility of the sample but may significantly influence the analysis results and the measurement of progressive motility. These include: concentration of sperm in the diluted sample, selection of fields for analysis, number of frames analyzed, framing rate and the threshold limits for VAP and STR that are used to “define” progressive motility. In our CASA system and in all systems used by SBS affiliate laboratories, a progressively motile sperm is defined as one which has a velocity (VAP) > 50 mic/sec and a straightness ratio (STR) > 75%.
Other laboratories may set up their CASA systems to have more or less strict requirements for a sperm to be considered progressive. For example, some labs use a much less strict definition (> 30 mic/sec for the VAP threshold and > 50% for the STR threshold). A sample that is reported to have 40% progressive motility by lab A (using the less strict definition) may be analyzed by lab B (using the more strict definition) and yield a very different value for progressive motility. This value may be well below 30% even though they are measuring the same exact sample and using the same sophisticated CASA system. For this reason it is important that comparison of reports of sperm motility as determined by CASA from two different labs be accompanied by a description of the analysis setups used. One must also recognize that there are other sources of variability which cannot be controlled. Even when two straws from the same ejaculate are thawed separately and analyzed separately on the same CASA system there is straw to straw variation in the measurements for progressive motility so we can expect as much as 5-7% variation in progressive motility between individual straws from the same ejaculate.
The Bottom Line
It is fortunate for those of us working with sperm that these cells are motile cells. We know that a dead sperm is non-motile and that when sperm are damaged it can affect the nature of their motility. We also know that motility is required for fertilization and that stallions whose sperm exhibit a high percentage of progressively motile sperm in the ejaculate tend to have higher fertility than those that have a low percentage of progressive motility. Measuring the quality of sperm motility before and after semen preservation (cooling or freezing) gives us an indication of how well that population of sperm survived the stresses of the process. However, motility is just one indicator of relative cell health that is easily observed and measured. Fertilization is a complex process involving numerous functional attributes of both sperm and oocyte. Some of these we can measure but many more we cannot. That is why some stallions with apparent normal sperm production and quality based on all the available laboratory tests we have are infertile. They are lacking in one or more of these functional attributes that we cannot measure or don’t even know exist. So a sample that has > 30% post-thaw motility is certainly not guaranteed to have any specific level of fertility. Also, a sample that has less than 30% motility may have quite good fertility if adequate sperm numbers are included in the dose.
So what value is there in measuring post-thaw sperm motility and striving to establish minimum standards for post-thaw quality for commercial semen? The value is in eliminating the majority of poor freezing stallions or poor quality samples that make their way onto the market. If the sample has very poor post-thaw motility it is more likely to have poor fertility than a sample that has very good post-thaw motility. The value of 30% is an arbitrary number and it is likely that a sample that is 25% progressively motile may be just as fertile as one that is 35%. A sample that has a lower percentage of motility after freezing and thawing has likely been more damaged than a sample that retains most of its motility through the process. By eliminating poor quality semen from commercial distribution we maximize the chances for success.
Use of objective systems for determining sperm motility such as CASA reduces the variability in motility estimates due to subjective bias. As an industry we should strive to adhere to minimum standards for post-thaw semen quality that is used in commercial breeding to protect all parties involved.





