 During those dog days of late summer and the end of the breeding season, most of us who work in equine reproduction are ready for a break. You are left with those difficult mares that didn’t settle earlier in the season and stallions that may be growing tired of breeding. Mares may not cycle as predictably or conceive as readily and stallions may be tougher to collect or exhibit a decrease in semen quality. One of the factors that can contribute to these problems is heat stress. As ambient temperatures rise, stallions and mares may experience disruptions to normal reproductive function as a result. This article will briefly describe some of the effects of heat stress on mare and stallion reproductive function.
During those dog days of late summer and the end of the breeding season, most of us who work in equine reproduction are ready for a break. You are left with those difficult mares that didn’t settle earlier in the season and stallions that may be growing tired of breeding. Mares may not cycle as predictably or conceive as readily and stallions may be tougher to collect or exhibit a decrease in semen quality. One of the factors that can contribute to these problems is heat stress. As ambient temperatures rise, stallions and mares may experience disruptions to normal reproductive function as a result. This article will briefly describe some of the effects of heat stress on mare and stallion reproductive function.
Stallions
Did you ever wonder why nature designed a system whereby an incredibly important organ like the testis, essential for species survival is suspended outside of the body protected only by a thin layer of scrotal skin? Stallion sperm are very temperature sensitive and normal spermatogenesis requires that the testis (site of sperm production) and epididymis (site of sperm maturation and storage) are maintained at about 3-4 degrees Celsius below body temperature, so a system evolved that allows for these special conditions to be maintained.
Exposure of the testes to elevated temperatures for prolonged periods may cause certain stages of developing sperm to be affected and die, resulting in a decrease in sperm numbers in the ejaculate or an increase in the percentage of morphologically abnormal sperm. Similarly, prolonged elevated scrotal temperature may cause abnormalities and dysfunction of sperm within the epididymis. This disruption in spermatogenesis and sperm quality may be evident within a few days after elevated scrotal temperature and may last for as long as 70 days after the cause of the increased temperature is removed. In addition to high ambient temperatures, an increase in scrotal temperature may be the result of a high fever or of swelling of the scrotum as a result of traumatic injury that insulates the testes.
Thermal regulation of the testis and epididymis is achieved by a coordination of mechanisms facilitated by the scrotum, the pampiniform plexus and the muscles supporting the testes. The testicular artery, located in the spermatic cord brings warm arterial blood to the testes. 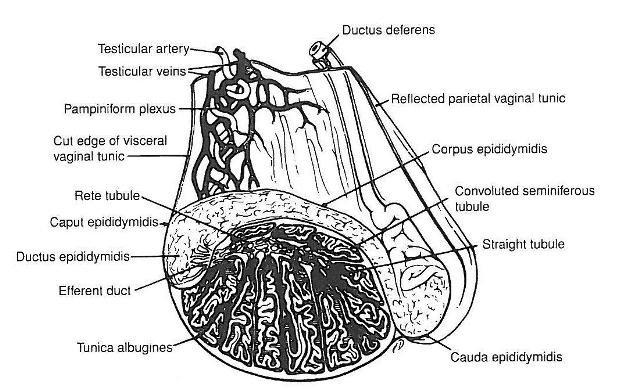 In a structure called the pampiniform plexus, the testicular artery is in intimate contact with a coiled network of testicular veins. The scrotum facilitates cooling of venous blood on the surface of the scrotum by evaporation of moisture from the scrotal skin. Within the pampiniform plexus this cool venous blood dissipates heat from the warm arterial blood so that the arterial blood reaching the testis is several degrees cooler than arterial blood within the body cavity and similar to the temperature just below the scrotal skin. Because of this heat exchange, the temperature of testicular tissue is similar to that of the cool venous blood. During hot weather muscles within the scrotal wall relax allowing the testicles to hang down further from the body cavity and allow for better cooling through the scrotal skin. In cold weather these muscles, and another within the spermatic cord contract bringing the testes closer to the body cavity to minimize heat loss.
In a structure called the pampiniform plexus, the testicular artery is in intimate contact with a coiled network of testicular veins. The scrotum facilitates cooling of venous blood on the surface of the scrotum by evaporation of moisture from the scrotal skin. Within the pampiniform plexus this cool venous blood dissipates heat from the warm arterial blood so that the arterial blood reaching the testis is several degrees cooler than arterial blood within the body cavity and similar to the temperature just below the scrotal skin. Because of this heat exchange, the temperature of testicular tissue is similar to that of the cool venous blood. During hot weather muscles within the scrotal wall relax allowing the testicles to hang down further from the body cavity and allow for better cooling through the scrotal skin. In cold weather these muscles, and another within the spermatic cord contract bringing the testes closer to the body cavity to minimize heat loss.
This system of thermal regulation is very efficient but clearly the degree of cooling is dependent to a great extent on the environmental temperature. In some hot climates during the summer months the ability to cool the testes through these mechanisms is inadequate and a seasonal, heat related decrease in semen quality is seen. Any management practices that provide a cool environment for stallions during the summer months may help to minimize negative effects on semen quality. Keeping stallions inside or in shaded areas during the day and providing fans will help promote dissipation of heat through the scrotal skin.
A note about exercise:
In a recent study conducted at Texas A&M University, Gordon and co-workers (2014) examined the effects of moderate exercise during summer months on subcutaneous scrotal temperature, semen parameters and hormone levels. Exercised stallions were worked outside at noon for 30 minutes a day on a motorized equine free exerciser for 4 days/week while control stallions that were not exercised were tied outside in close proximity to the exercised stallions. Exercise had no effect on the increase in rectal and scrotal temperatures that was observed for all stallions. Interestingly, even though rectal temperatures increased by 1.9C and scrotal temperatures increased by 0.8C, there were no differences seen in any of the semen parameters or hormone levels measured during the 12 weeks of the study or between the control and exercised stallions. The authors concluded that moderate exercise of breeding stallions during summer months would not affect breeding success.
Mares
So, what about the effect of heat stress on mare reproduction? Since all reproductive functions in the mare occur within the body, there is no similar complex heat exchange system in mares required to keep oocytes, ovaries or embryos cool. However, data from embryo transfer programs with competition mares and anecdotal reports from practitioners and breeders working in hot climates suggest that exercise induced heat stress or environmental heat stress may impair reproductive function in mares.
 Mares that were exercised outside between 1 and 3 PM for 30 minutes daily at ambient conditions in Texas where temperatures were >30C and humidity was >50% had a significantly lower embryo recovery rate when compared to non-exercised mares. (34% vs. 63%, Mortensen et al., 2009) Embryo quality also tended to be lower in exercised mares. In this study, exercise under these conditions caused a 2 degree Celsius increase in rectal temperature. In addition to lower embryo recovery rates, exercised mares also ovulated smaller follicles and had an increase in the time interval from prostaglandin administration to ovulation on the next heat. The published effect of heat stress on embryo recovery and quality in cattle is similar to that seen in the exercised mares in this study, however an embryo recovery rate of 63% seen in non-exercised mares held in the same environment suggest that exercise apart from environmental heat stress may be the culprit in the mare. It should be mentioned however that there are conflicting reports in the literature and studies by others have found no decrease in embryo recovery rates for embryo donor mares exercised in hot and humid climates (Pessoa et al., 2011 and Vasquez et al., 2010).
Mares that were exercised outside between 1 and 3 PM for 30 minutes daily at ambient conditions in Texas where temperatures were >30C and humidity was >50% had a significantly lower embryo recovery rate when compared to non-exercised mares. (34% vs. 63%, Mortensen et al., 2009) Embryo quality also tended to be lower in exercised mares. In this study, exercise under these conditions caused a 2 degree Celsius increase in rectal temperature. In addition to lower embryo recovery rates, exercised mares also ovulated smaller follicles and had an increase in the time interval from prostaglandin administration to ovulation on the next heat. The published effect of heat stress on embryo recovery and quality in cattle is similar to that seen in the exercised mares in this study, however an embryo recovery rate of 63% seen in non-exercised mares held in the same environment suggest that exercise apart from environmental heat stress may be the culprit in the mare. It should be mentioned however that there are conflicting reports in the literature and studies by others have found no decrease in embryo recovery rates for embryo donor mares exercised in hot and humid climates (Pessoa et al., 2011 and Vasquez et al., 2010).
In an attempt to determine if there is an effect of exercise apart from increased body temperature, Smith and co-workers (2012) exercised mares under conditions that did not raise rectal temperatures significantly and measured embryo recovery, blood flow to the ovary and the wall of the pre-ovulatory follicle as well as circulating levels of the stress-hormone, cortisol. Again, in this study embryo recovery rates were lower in exercised mares than non-exercised controls (43% vs. 67%). Also, despite increased blood flow through the ovarian artery, vascular perfusion (blood flow) of the follicular wall of pre-ovulatory follicles was reduced in the exercised mares. In those exercised mares, reduced perfusion was correlated with a reduced chance of recovering an embryo that cycle leading the authors to conclude that “exercise was detrimental to embryo recovery rate”. Another interesting aspect of this study suggests that this disruption of reproductive performance in the mare may be at the time of ovulation and conception rather than embryo development. In this study the exercised mares were split into two groups, one group was exercised daily throughout the reproductive cycle while the other group was exercised daily until the time of insemination and then rested until the day of embryo recovery attempt (day 7). Resting mares after insemination had no beneficial effect on recovery rates in the exercised mares.
Exercise and other stress factors can increase the circulating levels of stress hormones which can interfere with normal levels of reproductive hormones in mares at the level of the hypothalamus and ovary and lead to a disruption of ovarian function. Physical and psychological stress factors that may have a negative effect on reproductive performance of mares include heat, exercise, transportation, handling and change in environment. Management practices to minimize these stresses should be adopted to limit potential negative impact around the time of breeding, ovulation and the period between ovulation and embryo flushing for an ET donor or until early pregnancy is established for a mare bred to carry to term.
So, while moderate exercise in hot, humid conditions did not seem to contribute to impaired reproductive function of stallions, possibly because of the efficient system for thermoregulation of the testes, it does appear that exercise and other stress factors (including heat) may interfere with normal ovarian function in mares. Furthermore, extremely hot environmental temperatures that exceed the ability of the stallion to control scrotal temperature may result in impaired spermatogenesis and a subsequent decrease in semen quality.
_____________________________________________________________________________________
References:
Amann, R.P. (2011) Functional anatomy of the adult male. In: Equine Reproduction, 2nd edn, Ch 95 Editors: A.O. McKinnon, E.L. Squires, W.E. Vaala and D.D. Varner, Blackwell Publishing Ltd, Oxford
Gordon, R.K., Mawyer, J.D., Cavinder, C.A., et al. (2014) Effects of moderate exercise on semen parameters and serum LH and cortisol concentrations in stallions. Journal of Equine Vet.Sci., 34:65
Mortensen, C.J., Choi, Y.H., Hinrichs, K., Ing, N.H., Kraemer, D.C., Vogelsang, S.G. and Vogelsang, M.M. (2009) Embryo recovery from exercised mares. Anim. Reprod. Sci. 110, 237-244
Kelley, D.E., Gibbons, J.R., Smith, R., Vernon, K.L., Pratt-Phillip, S.E. and Mortensen, C.J. (2011) Exercise affects both ovarian follicular dynamics and hormone concentrations in mares. Theriogenology, 76, 615-622
Campbell, M.L.H., (2014) Embryo transfer in competition horses: Managing mares and expectations. Equine Vet. Educ. 26 (6) 322-327
Pessoa, M.A., Canninzza, A.P., Reghini, M.F.S. and Alvarenga, M.A. (2011) Embryo transfer efficiency of quarter horse athletic mares. J. Equine Vet. Sci. 31, 703-705
Vasquez, J.J., Garcia, A., Kass, P.H., Liu, I.K.M. and Ball, B.A. (2010) Influence of environmental temperature, exercise, semen type and ovulation characteristics on reproductive performance in a commercial embryo transfer program. Anim. Reprod. Sci. 121, 284-285





