A biofilm has been proposed to have a significant role in chronic infections in the horse. It has been suggested for over a decade that chronic uterine infections resistant to antimicrobials may be due to biofilm production. The involvement of a biofilm in cases of bacterial endometritis has not been clearly elucidated, but many reproductive specialists suspect a biofilm plays a significant role in infectious endometritis. In this article Dr. Ryan Ferris, a board certified theriogenologist from Colorado State University, explains the lifestyles of bacteria, how a biofilm is formed, how they protect bacteria and their implications on equine reproduction.
Lifestyles of Bacteria
Bacteria are capable of living in two different lifestyles, planktonic or biofilm states. Planktonic bacteria are single bacterial cells free flowing in suspension. Bacteria in this lifestyle utilize available nutrients for procreation. These individual cells are relatively susceptible to recognition and degradation by the host immune system, susceptible to changes in environment (dehydration, lack of nutrients, etc), and sensitivity to antibiotics. However, the planktonic cell paradigm does not accurately reflect the growth of bacteria in nature that are typically associated with a biofilm.
In the last several decades the biofilm state has been considered to be the more prevalent lifestyle with ~99% of the overall world bacterial biomass living in a biofilm. In natural environments these biofilms are invariably a multispecies microbial community harboring bacteria that stay and leave with purpose, share their genetic material at high rates and fill distinct niches within the biofilm.
How is a Biofilm Formed
 The first step in biofilm formation (photo right, biofilm formed in test tube) is migration and adherence to a surface. This is typically performed through the use of flagella and type IV pili in E. coli, P. aeruginosa, and K. pneumonia. Strep. equi subsp. zooepidemicus are non-motile and rely on movement from environmental or host factors. Individual bacteria will migrate (if capable) until other bacteria (same species or other) are encountered and micro-colonies start to form. At this point planktonic and biofilm lifestyles start to diverge, genes associated with flagella are down regulated and genes associated with polysaccharide production increase. This exopolysaccaride matrix forms the scaffold for the biofilm community.
The first step in biofilm formation (photo right, biofilm formed in test tube) is migration and adherence to a surface. This is typically performed through the use of flagella and type IV pili in E. coli, P. aeruginosa, and K. pneumonia. Strep. equi subsp. zooepidemicus are non-motile and rely on movement from environmental or host factors. Individual bacteria will migrate (if capable) until other bacteria (same species or other) are encountered and micro-colonies start to form. At this point planktonic and biofilm lifestyles start to diverge, genes associated with flagella are down regulated and genes associated with polysaccharide production increase. This exopolysaccaride matrix forms the scaffold for the biofilm community.
Biofilms Protect Bacteria
Clinically biofilms can cause significant difficulty for clinicians to eliminate these chronic infections once they are established. Bacteria within a biofilm are protected from the host immune system as white blood cells have reduced ability for movement and function, and the thick layer of exopolysaccaride (EPS) prevents antibodies from reaching bacteria deep within the biofilm. Biofilms protect bacteria from antibiotics by providing a diffusion barrier that decreases the amount of antibiotics that reach the protected bacterial colonies and creates a microenvironment that slows down the metabolism and therefore the replication rate of bacteria, which also makes them more resistant to antimicrobial agents.
Biofilm Effects on Mare Reproduction
The innate factors of antimicrobial resistance in bacterial biofilms have led to significant challenges in human medicine. It is estimated that 65% of hospital acquired infections are associated with biofilms and that treatments for biofilm based infections cost >$1 billion annually. In equine medicine, we have just started investigating the role of biofilms in chronic infections.
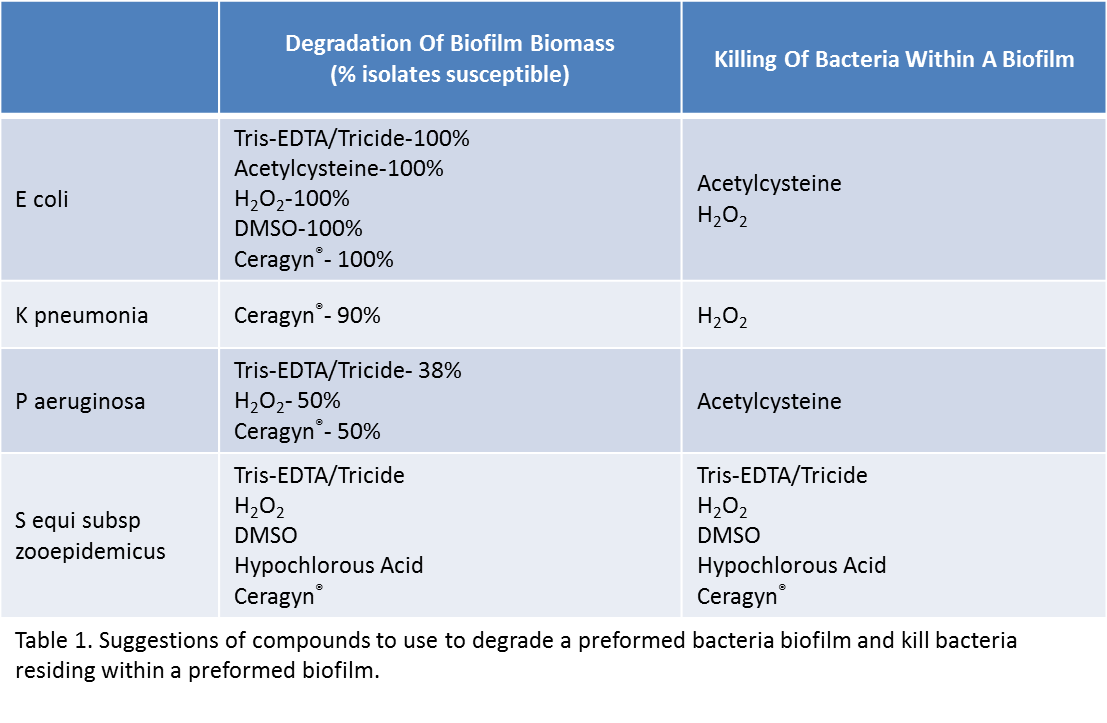
Our lab over the past two years has been able to show that ~80% of the bacteria isolated from the equine uterus are capable of forming a biofilm in vitro. Bacteria isolated from the equine uterus living in a biofilm state require ~1000x higher concentrations of antibiotics to kill the bacteria as compared to bacteria in a free-floating state. One way to increase the efficacy of our antibiotic treatment is to degrade the biofilm prior to antibiotic administration. A wide variety of compounds have been suggested to degrade biofilm such as tris-EDTA, DMSO, hydrogen peroxide and acetylcysteine. Unfortunately, not one compound was capable of degrading a preformed biofilm in every one of the four most common bacteria isolated from the equine uterus (Table 1). For example DMSO was able to degrade a preformed biofilm in E. coli and Strep. zooepidemicus but unable to affect isolates of P. aeruginosa and K. pneumonia .
In conclusion biofilms may play a role in cases of chronic bacterial endometritis. For effective treatment of a bacterial infection involving a biofilm identification of the causative organism is required. Once the causative organism is identified an appropriate treatment plan involving agents to disrupt the biofilm and antibiotics to kill the bacteria can be developed. Further work is ongoing to determine the role of biofilm in cases of bacterial endometritis. Hopefully this work will lead to improved diagnostic and therapeutics for mares with chronic bacterial endometritis.
If you like this article you may be interested in:
The Facts About Frozen Semen and Post Breeding Uterine Fluid





